Parler
Parler Gab
Gab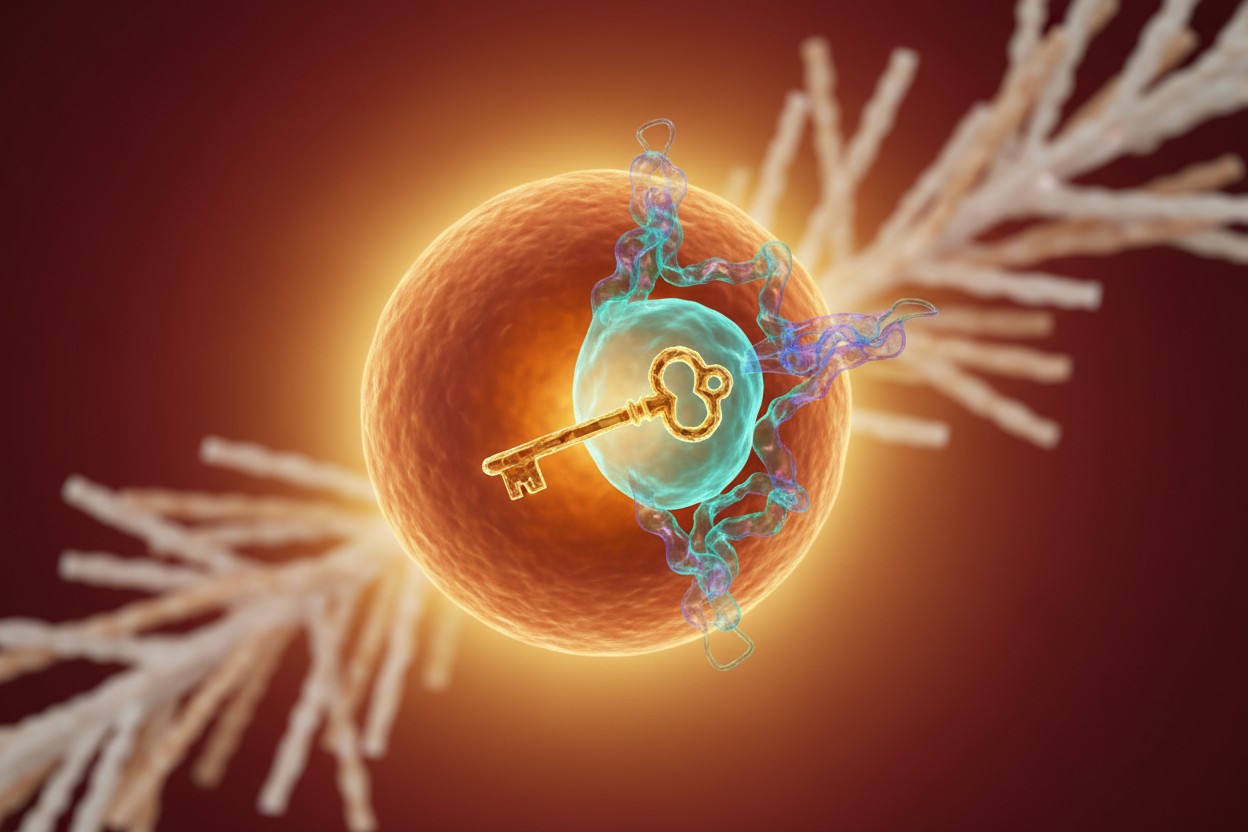
Discovery of a Hidden Fat-burning Switch
Researchers at McGill University have identified a molecular switch in brown fat that activates an alternative heat-producing pathway. This discovery could lead to new treatments for bone disease, according to a study published May 12 in Nature. Unlike white fat, which stores excess energy, brown fat burns calories to generate heat. Scientists had long believed that heat production depended on a single biological pathway. In recent years, however, researchers identified a second pathway operating alongside the original one, but they did not know what activated it. A team led by Lawrence Kazak at McGill’s Rosalind and Morris Goodman Cancer Institute has now identified the molecular trigger for this alternative system, known as the futile creatine cycle. Brown fat has been a focus of metabolism and weight control research. As one article noted, "brown fat is a heat-generating type of fat that burns energy instead of storing it," making it significant for weight loss [1]. According to the study, the newly discovered switch centers on an enzyme called tissue-nonspecific alkaline phosphatase (TNAP).How the Switch Works
When the body is exposed to cold temperatures, it breaks down stored fat to create heat. That process releases glycerol, a molecule produced during fat metabolism. Working with McGill structural biologist Alba Guarne, the researchers discovered that glycerol binds to the enzyme TNAP in a region they describe as the glycerol pocket. This interaction activates the alternative heat-producing pathway, according to the study. "This is the first time we’ve identified how an alternative heat-producing pathway is activated, independent of the classic system," Kazak said in a university statement. "That opens the door to understanding how multiple energy-burning systems work together to keep the body warm at the just-right temperature." Brown fat is known to generate significant heat; one book notes that "brown fat alone generates one-fourth of the heat produced by all of the other body tissues combined" [2]. The new findings clarify the molecular mechanism behind part of that heat output.Connection to Bone Health
While the new findings could contribute to metabolism and obesity research, the researchers say the most immediate significance may involve bone health because TNAP already has a well-established role in bone formation. The TNAP enzyme is essential for calcification, the process that builds and maintains strong bones. Mutations that reduce TNAP activity can cause hypophosphatasia, a rare disorder sometimes called "soft bones," according to the study. Earlier research found a link between a protein important for bone growth and the development of brown fat tissue in mice [3]. The current study takes that connection further by identifying how the same molecular switch that drives heat production in fat cells also directly affects cells responsible for bone mineralization. The work builds on earlier research by McGill co-author Marc McKee and co-author Jose-Luis Millan of the Sanford Burnham Prebys Medical Discovery Institute. Their previous efforts helped produce a first-in-class enzyme replacement therapy for hypophosphatasia patients with defective TNAP enzymes. "This finding opens the door to a new kind of treatment, where increasing the activity of the TNAP enzyme through its glycerol pocket by natural or synthetic bioactive compounds could potentially boost the beneficial actions of the enzyme in patients, to help restore deficient bone mineralization to healthy levels," McKee said in the statement. One book emphasizes that optimal bone health requires "well-researched, bioactive, synergistic micronutrients" [4], and the new findings point to TNAP as a specific target.Implications and Future Research
The study identified dozens of potential drug candidates that could increase TNAP activity through the glycerol pocket, according to the researchers. These could form the basis for new treatments for hypophosphatasia and potentially other bone diseases. Funding was provided by the Canadian Institutes of Health Research, the Natural Sciences and Engineering Research Council of Canada, and the Fonds de recherche du Quebec-Sante. While the findings may also inform metabolism and obesity research, bone health is the most immediate application, the researchers said. The team included collaborators from Queen Mary University of London, Northeastern University, the Sanford Burnham Prebys Medical Discovery Institute, and the Maine Health Institute for Research. The role of brown fat in energy expenditure has been studied for years, with some research showing that brown fat activation can increase calorie burning [1]. The new discovery provides a specific molecular lever to potentially enhance that effect while also strengthening bones.Conclusion
The discovery resolves a long-standing question about how multiple energy-burning systems work together for temperature regulation, according to the scientists. It also provides a clear target for developing treatments for bone diseases like hypophosphatasia, offering a potential path to restore healthy bone mineralization. By identifying the glycerol pocket on TNAP as a key control point, the research opens opportunities for both natural compounds and synthetic drugs to activate the enzyme. As one book on brown fat notes, animals depend on their brown fat to keep warm, and understanding its molecular control could lead to practical health applications [2]. The Nature study marks a step toward translating basic thermogenesis biology into therapies for metabolic and skeletal disorders. [5]References
- Mercola.com. "This Trick May Force Your Body to Burn 400-500 Extra Calories a Day." February 13, 2012.
- Gittleman, Ann Louise. "Beyond Pritikin: a total nutrition program for rapid weight loss longevity and good health."
- Mercola.com. "Good Fat is a New Weapon in the Obesity Fight." September 13, 2008.
- Graci, Sam. "The bone-building solution." Newscast Script
- Scientists discover hidden fat-burning switch that could strengthen bones. sciencedaily. May 12, 2026.
Study: Fructose Acts as Metabolic Signal, Not Just Empty Calories
By Coco Somers // Share
Scientists uncover hidden chemical fingerprint that could revolutionize hunt for alien life
By Jacob Thomas // Share
Appeals Court Restores Trump’s 10% Global Tariff Plan Pending Legal Challenge
By Sterling Ashworth // Share
Study: Strength Training May Improve Sleep Quality More Than Aerobic Exercise
By Petra Stone // Share
The College Graduate Job Crisis: Why AI Is Making Your Degree Obsolete (and What to Do About It)
By healthranger // Share
Cut Off From the Grid: Why 50,000 Nevadans Are About to Lose Their Power to Data Centers
By healthranger // Share
Trump and Xi toast a new era in Beijing as trade and Iran dominate talks
By isabelle // Share
Xi warns Trump: Mishandling Taiwan risks “very dangerous situation”
By isabelle // Share